Project Management
Wish you could see the health of your project on one page? Each project has a dashboard home-page that summarizes all of the information regarding the project into a comprehensive, easily digestible form that provides a “one-stop-shop” for people interested in understanding the overall status of the project at a glance. It contains summary-level information for all types of artifact (requirements, test cases, incidents, etc.) that you can use to drill-down into the appropriate section of the application.
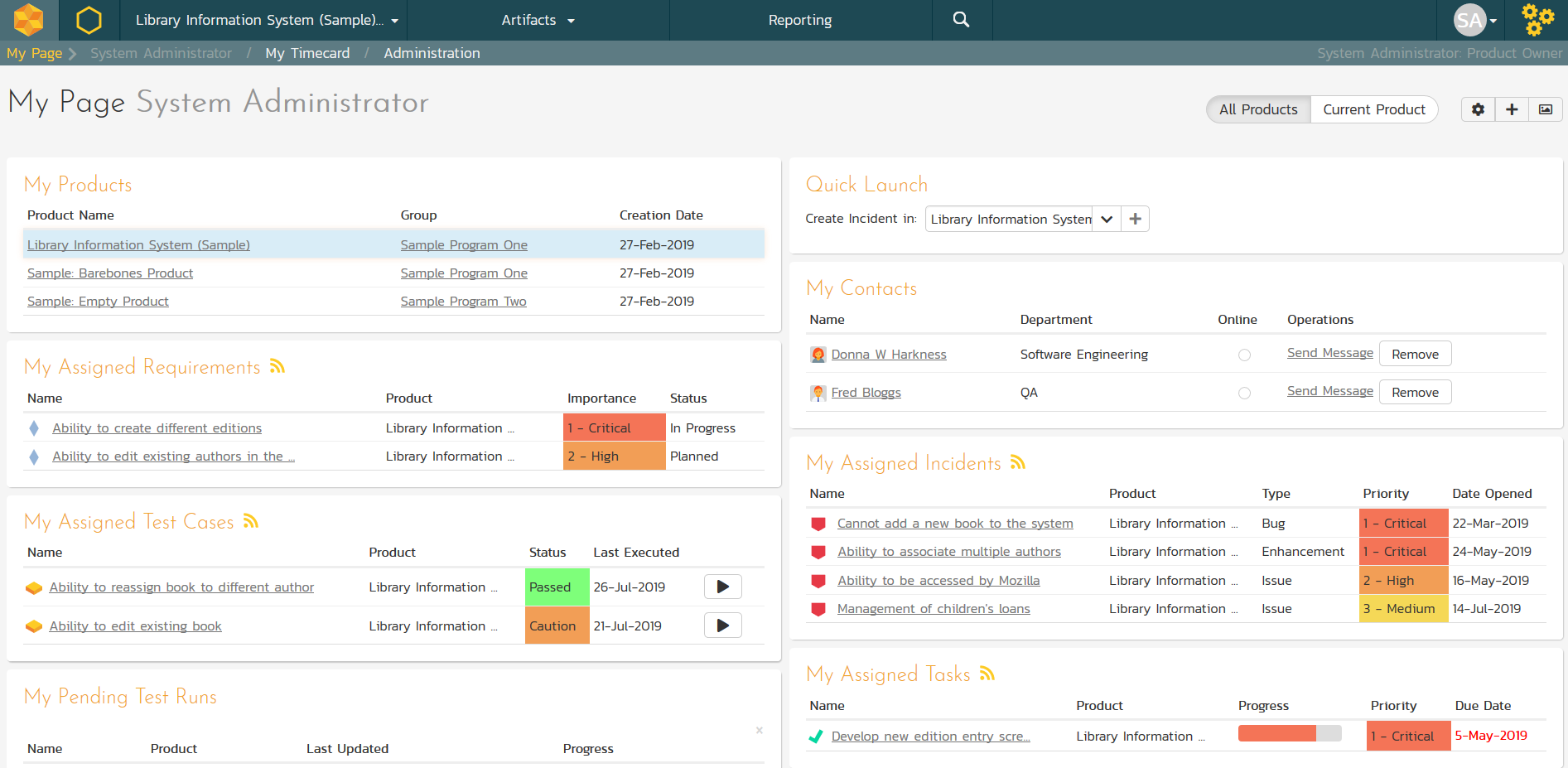
Personalized Home Page
When you first login to SpiraTeam, you have a personalized ‘dashboard’ of all your key information, consolidated onto a single page for you to take immediate action. The system provides RSS feeds of your assigned items that you can subscribe to using a RSS newsreader of your choice.
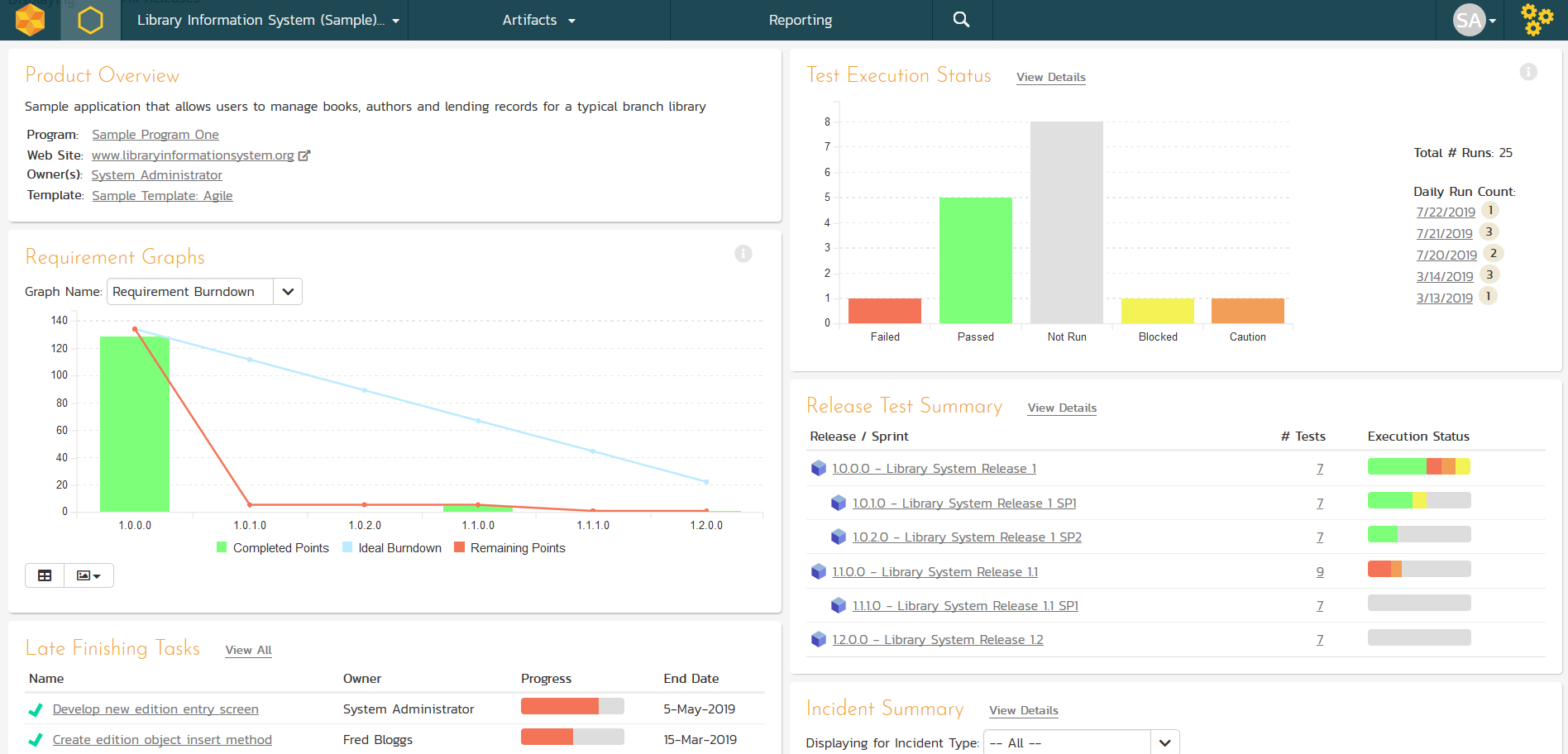
Project Dashboards
Inside SpiraTeam, each project has a dashboard home-page that summarizes all of the information regarding the project into a comprehensive, easily digestible form. It provides a “one-stop-shop” for people interested in understanding the overall status and health of the project at a glance.
The project dashboard contains summary graphs and metrics for planning, estimating and monitoring the progress of your project. All of the graphs provide the ability to quickly drill-down into the appropriate section of the application and understand the information behind the metric. SpiraTeam includes standard graphs such as task burndown, requirements test coverage, and test case execution status.
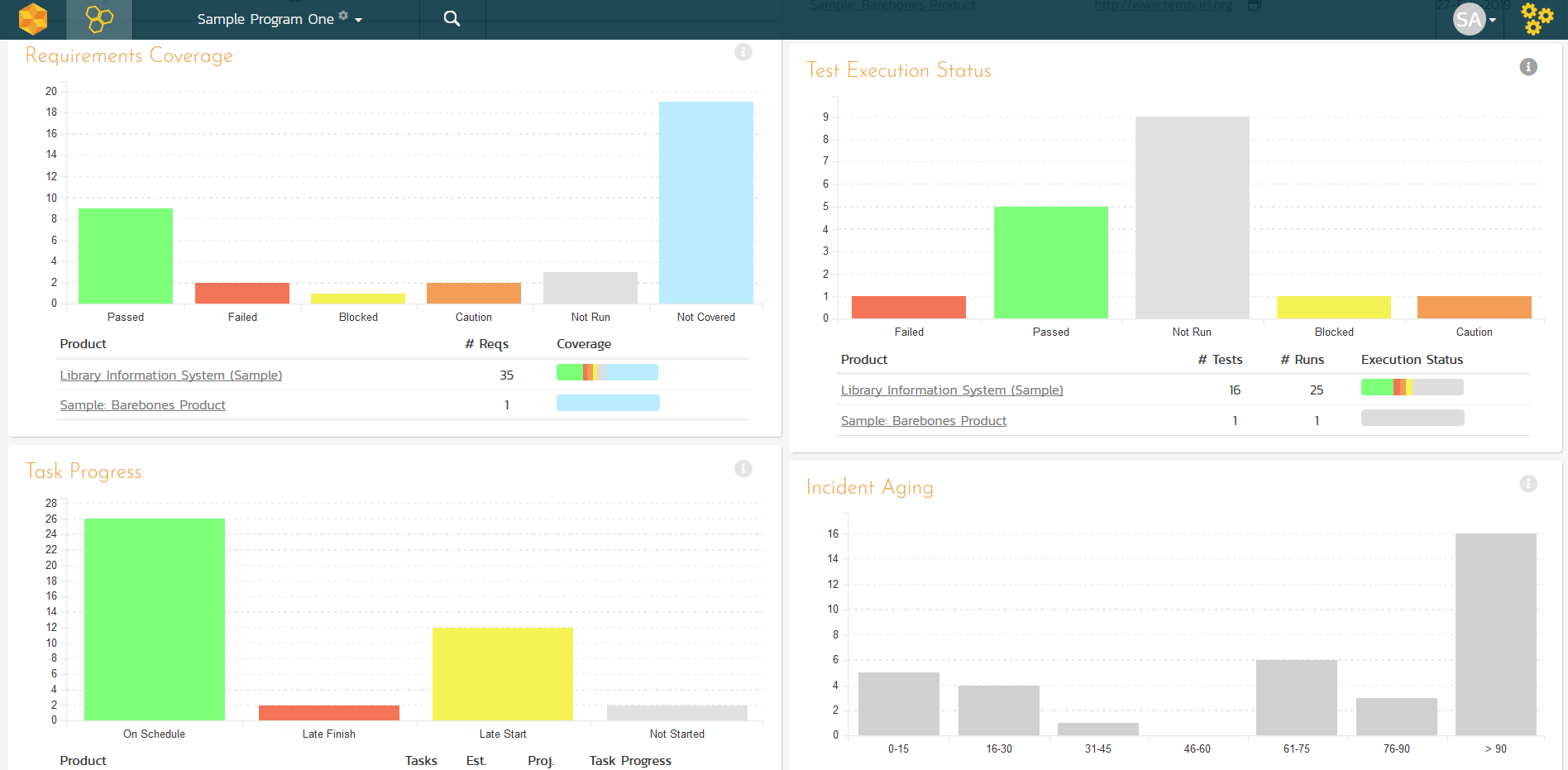
Cross-Project Reporting
Projects in SpiraTeam are organized into Project Groups. This lets you report on all of the projects in a specific organization, customer or division. You can see the aggregate status of the group as whole and in addition, compare the relative health of the different projects in the group:
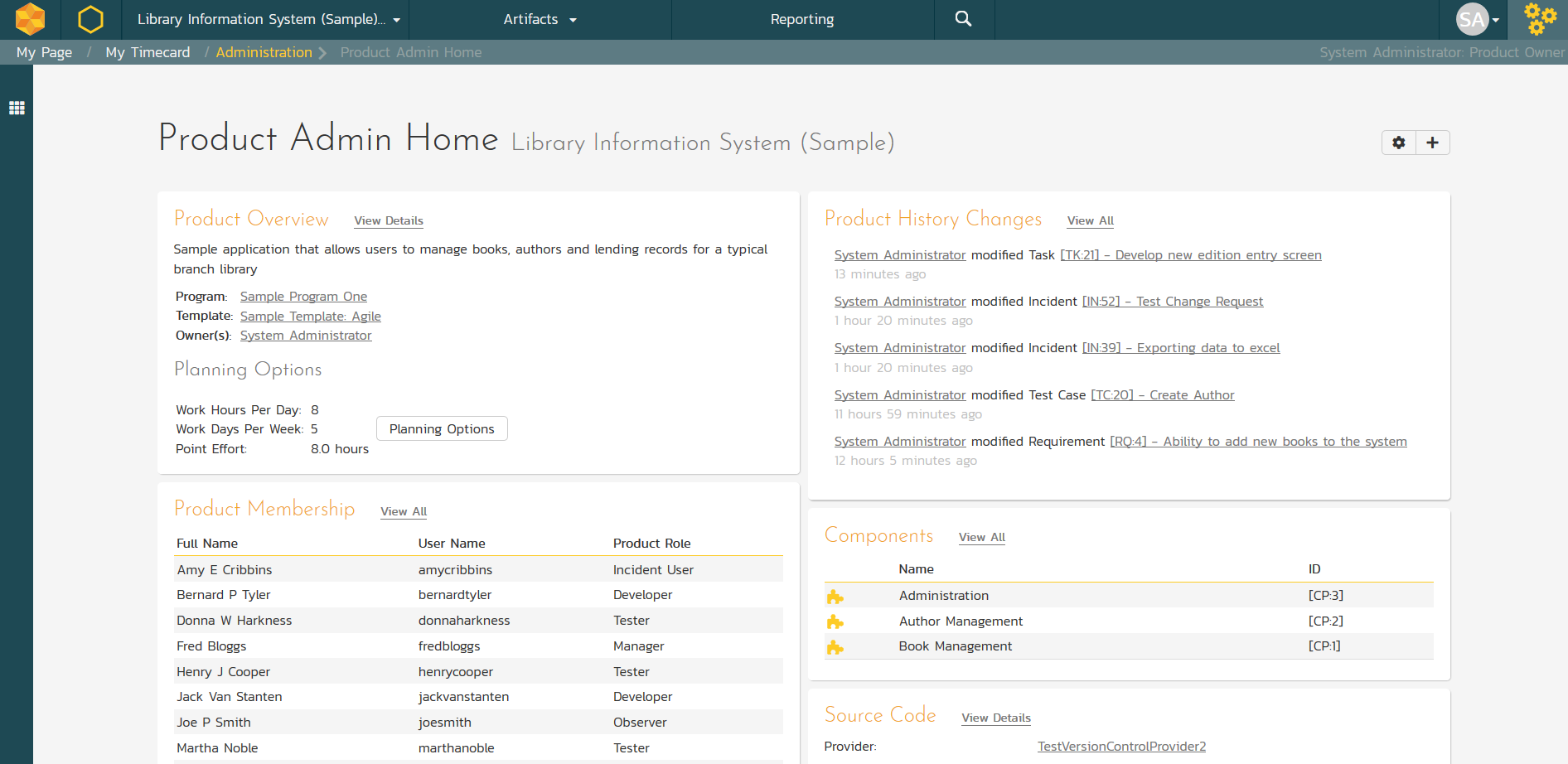
Project Administration
Managing your projects is easy with SpiraTeam’s easy to use, administrative portal. It lets you see all of the members of your project, their recent activity, and key modules such as project Components, data synchronization with other tools, and source code repositories.
Try SpiraTeam free for 30 days, no credit cards, no contracts
Start My Free TrialAnd if you have any questions, please email or call us at +1 (202) 558-6885